Description
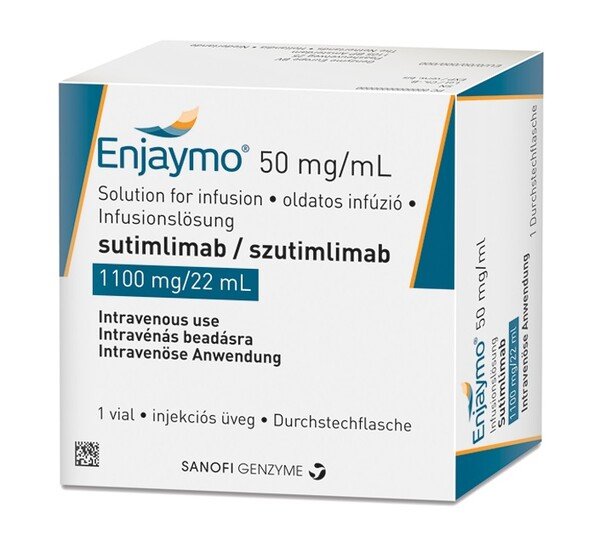
Sutimlimab is indicated to decrease the need for red blood cell transfusion due to hemolysis (red blood cell destruction) in adults with cold agglutinin disease (CAD).
The most common side effects include respiratory tract infection, viral infection, diarrhea, dyspepsia (indigestion), cough, arthralgia (joint stiffness), arthritis, and swelling in the lower legs and hands.[3] In two phase 3 studies (n=66), 29% of patients experienced infusion-related reactions, including shortness of breath, rapid heartbeat, nausea, flushing, headache, hypotension, chest discomfort, pruritis, rash, injection site reaction, and dizziness.
ENJAYMO (sutimlimab-jome) injection, for intravenous use Initial U.S. Approval: 2022
See full prescribing information : Click Here
Need Help Getting Medicine? Here’s How We Can Assist You.
Brand Name “ENJAYMO” or Generic Name “sutimlimab-jome” can be imported for personal use under “Named Patient Program” treatment in Delhi, Kolkata, Surat, Jaipur, Noida, Gurgaon (Gurugram), Punjab, Chandigarh, Bhubaneswar, Arunachal Pradesh, Assam, Manipur, Meghalaya, Mizoram, Nagaland, Tripura and Sikkim, Noida, Kanpur, Lucknow, Dehradun, Shimla, Ahmedabad, Jodhpur, Mumbai, Jaipur, Aurangabad, Pune, Bangalore, Hyderabad, Chennai, Visakhapatnam, Coimbatore, Andhra Pradesh, Karnataka, Kerala, Lakshadweep, Puducherry, Tamil Nadu, Telangana, India. Contact us at support@southdelhipharma.net, southdelhipharma@gmail.com or you can call at 9891296838 or WhatsApp at 9891296838